Expert IRB Review, Consulting, IRB Auditing, AI Assisted Grant Writing and IRB Development
IRB Review & Consulting
Guiding you through IRB processes and requirements.
Comprehensive audits for research compliance and ethics.
Grant writing to meet your regulatory needs.
Auditing
Grant Writing
IRB Services Overview
IRB services including approvals, review of changes to protocol, Non-Human Research, Exempt Review, and Expedited Review.
We offer IRB auditing services. including IRB and GCP compliance while enhancing the protection of research participants at a low price.






Our dissertation and education programs equip researchers with knowledge about IRB processes and ethical research practices.
or
Book an Appointment Below!
Why Choose Compita Consulting for Your IRB Needs?
We offer over 30 years of IRB review services from investigator-initiated/sponsored research, which aims to foster a consultative relationship with investigators. During the pre-review process, an experienced, qualified Research Compliance Advisor (RCA) guides the investigator/sponsor through the submission and review process, assisting with protocol and informed consent form development, education or auditing, and completion of IRB application by our seasoned professional when needed.


How IRBs Protect Human Research Participants
Informed Consent for Research:
What to Expect
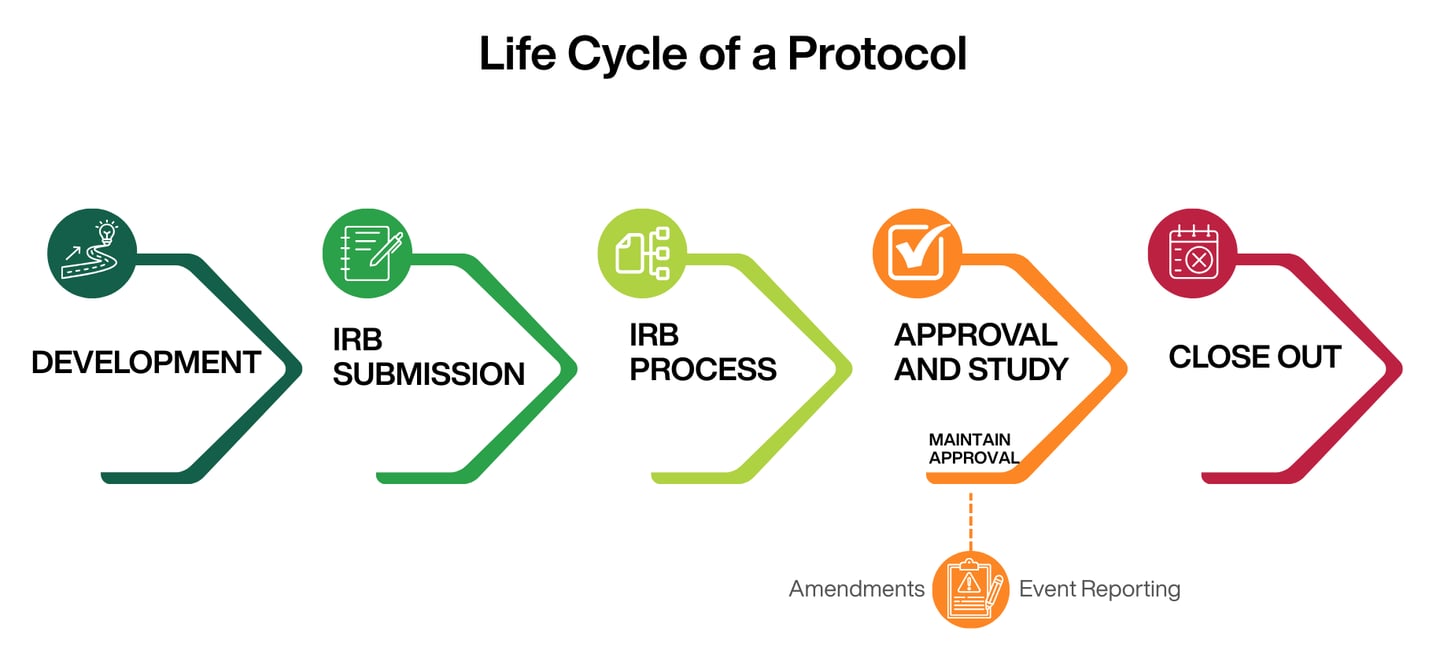
Frequently Asked Questions
What are your services?
Compita Consulting, LLC, operates an institutional review board that provides independent IRB reviews, consulting, auditing, and IRB for dissertation research.
How does your product/service work?
We offer a variety of services. We offer IRB review, consulting services, dissertation assistance (coaching, editing, development), and education needs to help our customers. Our process is efficient, straightforward, and convenient, and you will expedite your research approval.
How much does it cost?
Our pricing varies depending on the level of review or the level of assistance you need. We offer a variety of options to fit your needs and budget. Please visit our pricing page/contact us for more information.
Do you offer IRB Review of Clinical Trials?
We don't currently offer Full Board Review (Coming Soon). However, we do offer exempt, expedited or non-human research. Additionally, we offer Consulting, Dissertation Assistance, Education and Audit and Monitoring services which might be helpful.
How can I contact you?
For immediate assistance, contact Dr. Andrea Dadas at (347) 671-9867, call the office at (785) 410-8003, or book a meeting. We are always happy to answer your questions.
Where can I learn more about you?
You can explore other sections of this website, such as the "About Us" page or our blog, to learn more about our company, mission, and team.
Client Feedback
Discover how our services have positively impacted research compliance and ethics.
Compita Consulting provided invaluable guidance, ensuring our research met all ethical standards and received IRB approval.
Rochester, MN
The consulting from Compita Consulting's IRB Services was crucial for our project’s success and compliance with regulations. Their expertise made a significant difference in our research process.
John T.
Anaheim, CA
★★★★★
★★★★★
Sandy B.
Compita Consulting operates an Independent Institutional Review Board (IRB) that provides ethical review at an affordable rate. We ensure that community-based organizations, small organizations, and independent investigators are educated in Human Research Protections and conduct research that meets ethical guidelines and federal regulations. We provide experienced Research Compliance Advisors to support every stage of your study, from start-up to close-out, with efficient reviews, fast turnaround times, and assistance preparing IRB applications, informed consent documents, and minor modifications. Please contact us at (347) 671-9867
IRB Services Parent Organization# IORG0011844, IRB Services# IRB00014028
Compita Consulting IRB Services
Expert Guidance for Human Research Protection
Contact us
© 2026, Compita Consulting, LLC, all rights reserved.


